Pelvic Organ Prolapse Surgery
JOIN OUR NEWSLETTER
Receive custom tools to help you manage your condition and get the latest in bladder and bowel health from NAFC!
For those contending with prolapse issues, reconstructive surgery for Pelvic Organ Prolapse (POP) is a viable option. Ultimately, the purpose of the surgery is to correct the anatomy as well as provide better bowel, bladder, and vaginal function.
ANATOMY
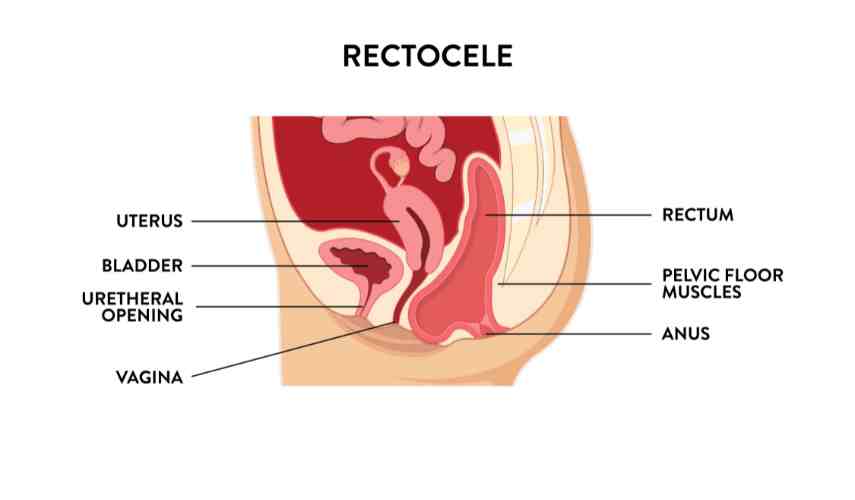
Take a moment or two to reacquaint yourself with the female anatomy so you can understand how the components are interwoven and connected.
Prolapse or support problems can affect one or multiple organs of the pelvis. Weakness of the front-side vaginal wall near the bladder results in a cystocele, often called a “dropped bladder”.

Weakness of the vaginal ceiling results in uterine prolapse, known as an enterocele.

Weakening of the backside vaginal wall near the rectum results in a rectocele.
Surgery
Prolapse repairs can be done transvaginally, abdominally, laparoscopically, and/or robotically (i.e., when a scope is placed through the belly button). Correcting all support defects is paramount in the surgical approach to POP. Abdominal repairs are thought to have higher success rates at the cost of increased morbidity when performed through a large incision.
Because of less than optimal success rates with traditional repairs, pelvic surgeons are constantly looking for new surgeries to approach this problem. Many surgeons are using vaginal grafts (made of synthetic and biologic materials) in attempts to improve long-term success rates; however, limited research has been done to prove that these methods improve results without increasing complications. While use of permanent mesh that is placed vaginally may improve vaginal support, many women have good results with repairs that are performed using their own tissues.
Research is currently being done to determine if the benefits of using mesh grafts in POP surgical repairs for greater durability sufficiently outweigh the risks of undesirable adverse consequences. Limited research to date indicates that women who are older, smoke, are diabetic, or have had a hysterectomy are at higher risk for these more common complications.
For women who never plan on having sexual intercourse again, there are simple transvaginal surgeries that have nearly a 100% success rate. In these techniques the vagina is sewn shut and shortened so that it no longer prolapses. After these surgeries, vaginal intercourse is impossible. These techniques are ideally suited for the elderly patient with multiple medical problems that would otherwise place her at increased risk with a reconstructive approach.
Evaluation
Pelvic organ prolapse (POP), the herniation of the pelvic organs to or beyond the vaginal walls, in women is diagnosed using pelvic examination. A medical history is also important to elicit prolapse-associated symptoms, since treatment is generally indicated only for symptomatic prolapse.
The Baden-Walker scale is often used to evaluate the severity of the condition from 0 to 4. Another evaluation tool, the Pelvic Organ Prolapse–Quantification (pelvic organ prolapse-Q; stages 0 through IV) is an international system that is more complex and involves taking several measurements. Both produce reliable results, giving your physician and you the information to proceed.
Recovery
Since POP surgery is fairly invasive, recovery will take time. Among the things to anticipate:
Postoperative Pain – Pain medication can be controlled and delivered by the patient via an intravenous patient-controlled analgesia (PCA) pump or by injection (pain shot) administered by the nursing staff. You may experience minor transient shoulder pain (1-2 days) related to the carbon dioxide gas used to inflate your abdomen during the laparoscopic surgery.
Bladder Spasms – Bladder Spasms are commonly experienced as a moderate cramping sensation in the lower abdomen or bladder and are common after colposuspension. These spasms are usually transient and often decrease over time. If severe, medications can be prescribed by your doctor to decrease the episodes of these spasms.
Nausea – You may experience transient nausea during the first 24 hours following surgery, which can be related to the anesthesia. Medication is available to treat persistent nausea.
Urinary Catheter – You can expect to have a urinary catheter (i.e., Foley), which is placed in the operating room under anesthesia, draining your bladder for approximately 1-2 days after the surgery. It is not uncommon to have blood-tinged urine for a few days after your surgery.
Vaginal Packing – Vaginal gauze packing is routinely placed at the end of the operation while the patient is under anesthesia. This packing will typically be removed the next day.
Diet – You can expect to have an intravenous catheter (IV) in for 1-2 days. (An IV is a small tube placed into your vein so that you can receive necessary fluids and stay well hydrated. In addition, it provides a route to receive medication.) Most patients are able to tolerate clear liquids the first day after surgery, and a regular diet the following day. Once on a regular diet, pain medication will be administered by mouth instead of by IV or shot.
Fatigue – Fatigue is common and should start to subside in a few weeks.
Incentive Spirometry – You will be expected to do some very simple breathing exercises to help prevent respiratory infections by using an incentive spirometry device. These exercises will be explained to you during your hospital stay. Coughing and deep breathing is an important part of your recuperation and helps prevent pneumonia and other pulmonary complications.
Ambulation – On the day after surgery it is very important to get out of bed and begin walking with the supervision of your nurse or family member to help prevent blood clots from forming in your legs. You can expect to have SCD’s (sequential compression devices) along with tight, white stockings on your legs to prevent blood clots from forming in your legs while you are lying in bed.
Hospital Stay – Length of hospital stay for most patients is 1-2 days.
Constipation- You may experience sluggish bowels for several days to a week after surgery. Suppositories and stool softeners may be given to help with this problem. Taking 1 tsp. of mineral oil and milk of magnesia at home will also help to prevent constipation.
Is Surgery For Me?
With the guidance of your physician, only you can make the final decision on whether to go through with surgery. You must weigh the risks versus the rewards and try to envision your life after surgery. Look past the short-term pain associated with the procedure and try to imagine the impact on your lifestyle.
Once you review the pros and cons with your physician and understand the procedure to the best of your ability, only then can you make the decision right for you.
RELATED ARTICLES

From Postpartum to Postmenopause: The Conversations Women Need to Have
In this episode of Life Without Leaks, we’re joined by Dr. Barbara Frank of Harvard Medical School and Brigham and Women’s Hospital, along with Alex Fennell, co-founder of Attn: Grace, an innovative manufacturer of incontinence products.
Together, we explore the realities of bladder leaks and pelvic floor health during pregnancy, postpartum recovery, perimenopause and menopause, and we discuss why more women are finally beginning to talk more openly about their experiences.
The conversation covers:
• The connection between hormones, menopause and bladder leaks
• Why pelvic floor therapy is having a major moment
• The surprising relationship between pelvic floor tension and incontinence
• How products can impact skin health, comfort and UTIs
• The truth about hormone replacement therapy
• Why community and conversation matter so much in women’s health
Alex also shares the story behind Attn: Grace and the company’s mission to create cleaner, plant-based incontinence products designed to support women with comfort, dignity and confidence.
Whether you’re navigating postpartum recovery, menopause symptoms or simply looking to better understand pelvic health, this episode offers expert insights, practical advice and an encouraging reminder that no one should feel alone in these experiences.

Ask The Expert: From A Doctor’s Perspective What Are Some Common Questions You Hear?
If you’ve ever sneezed and held your breath hoping for the best, you’re not alone. Bladder leaks, pelvic floor issues, and the constellation of symptoms that go with them are incredibly common — and incredibly under-discussed. We sat down with two of our favorite experts who are changing the bladder leak conversation: Dr. Barbara Frank, Attn: Grace medical advisor and board certified OB/GYN, and Dr. Sara Reardon, pelvic floor physical therapist (and the woman your pelvic floor has been waiting to meet). They took turns asking each other the questions their patients ask most — and answered them with the same candor they bring to the exam room. They cover everything from how to prepare for postpartum, to what question to ask your own pelvic floor PT, to how to manage incontinence during menopause. This is a knowledge drop you don’t want to miss.

How Aging Changes Bladder Control — What Most People Don’t Expect
As an RN who’s worked in geriatrics for over 14 years, I know first hand that changes in bladder control can be a common part of aging. However, what I’ve also learned is that the treatment and management of these changes differ depending on whether they are due to aging or other conditions.
If you’re experiencing more frequent trips to the bathroom or sudden, intense urges to urinate, you might be wondering what is going on and what you can do about it. Let’s talk through some of the changes that might be going on in your body and how you can be better prepared to manage your bladder control.

From Postpartum to Postmenopause: The Conversations Women Need to Have
In this episode of Life Without Leaks, we’re joined by Dr. Barbara Frank of Harvard Medical School and Brigham and Women’s Hospital, along with Alex Fennell, co-founder of Attn: Grace, an innovative manufacturer of incontinence products.
Together, we explore the realities of bladder leaks and pelvic floor health during pregnancy, postpartum recovery, perimenopause and menopause, and we discuss why more women are finally beginning to talk more openly about their experiences.
The conversation covers:
• The connection between hormones, menopause and bladder leaks
• Why pelvic floor therapy is having a major moment
• The surprising relationship between pelvic floor tension and incontinence
• How products can impact skin health, comfort and UTIs
• The truth about hormone replacement therapy
• Why community and conversation matter so much in women’s health
Alex also shares the story behind Attn: Grace and the company’s mission to create cleaner, plant-based incontinence products designed to support women with comfort, dignity and confidence.
Whether you’re navigating postpartum recovery, menopause symptoms or simply looking to better understand pelvic health, this episode offers expert insights, practical advice and an encouraging reminder that no one should feel alone in these experiences.



